 
Further radial nodes become evident in higher d -orbitals ( 5 d, 6 d, and 7 d) but fewer nodes in lower d -orbitals ( 3 d. 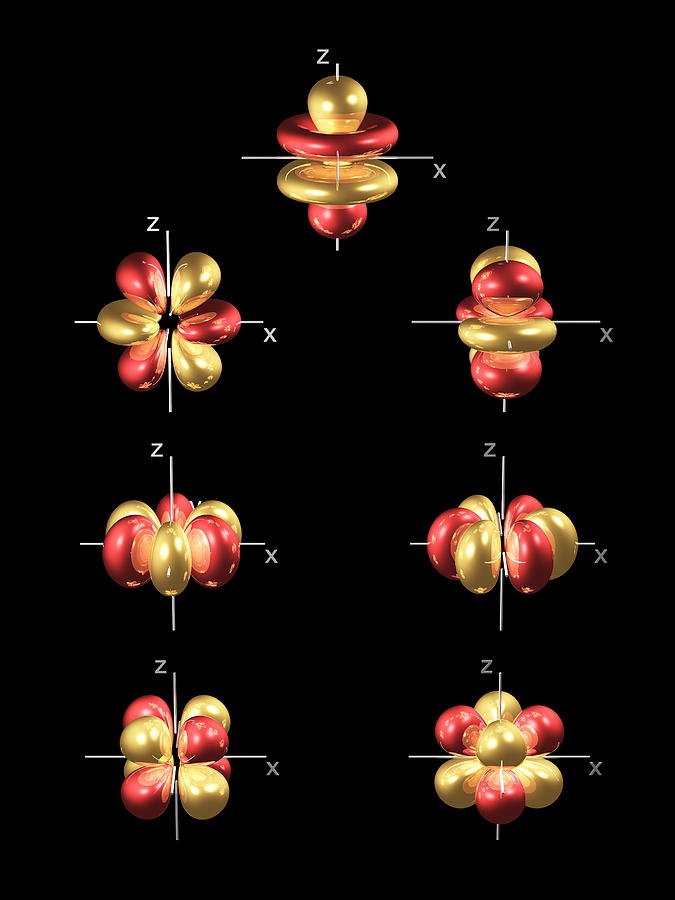
It is independent of the values of the other quantum numbers. Therefore, the 4 d -orbitals each have (4 - 3) 1 radial node, as shown in the above plot. A Radial Node can be defined as the spherical area near the elements nucleus where there is a. The spin quantum number describes the direction in which the electron is spinning. The total number of angular nodes found in the 4d orbital is two. In addition, a dumbbell-shape 4 d t2g recombined dxz dyz orbital ordering on the Ru sublattice is obtained owing to the on-site Coulomb interaction U associated with the elongated RuO 6. The correct option is B 6 h 2For d-orbital, l 2where l Azimuthal quantum numberOrbital angular momentum is given byL h 2l(l 1)Putting the value of l, we getL h 22(2 1) 6 h 2. There are four quantum numbers, namely, the principal quantum number, represented by $ n $, orbital angular momentum or Azimuthal quantum number, represented by $ l $, magnetic quantum number, represented by $ \right) $ integral values from $ - l $ to $ l $. 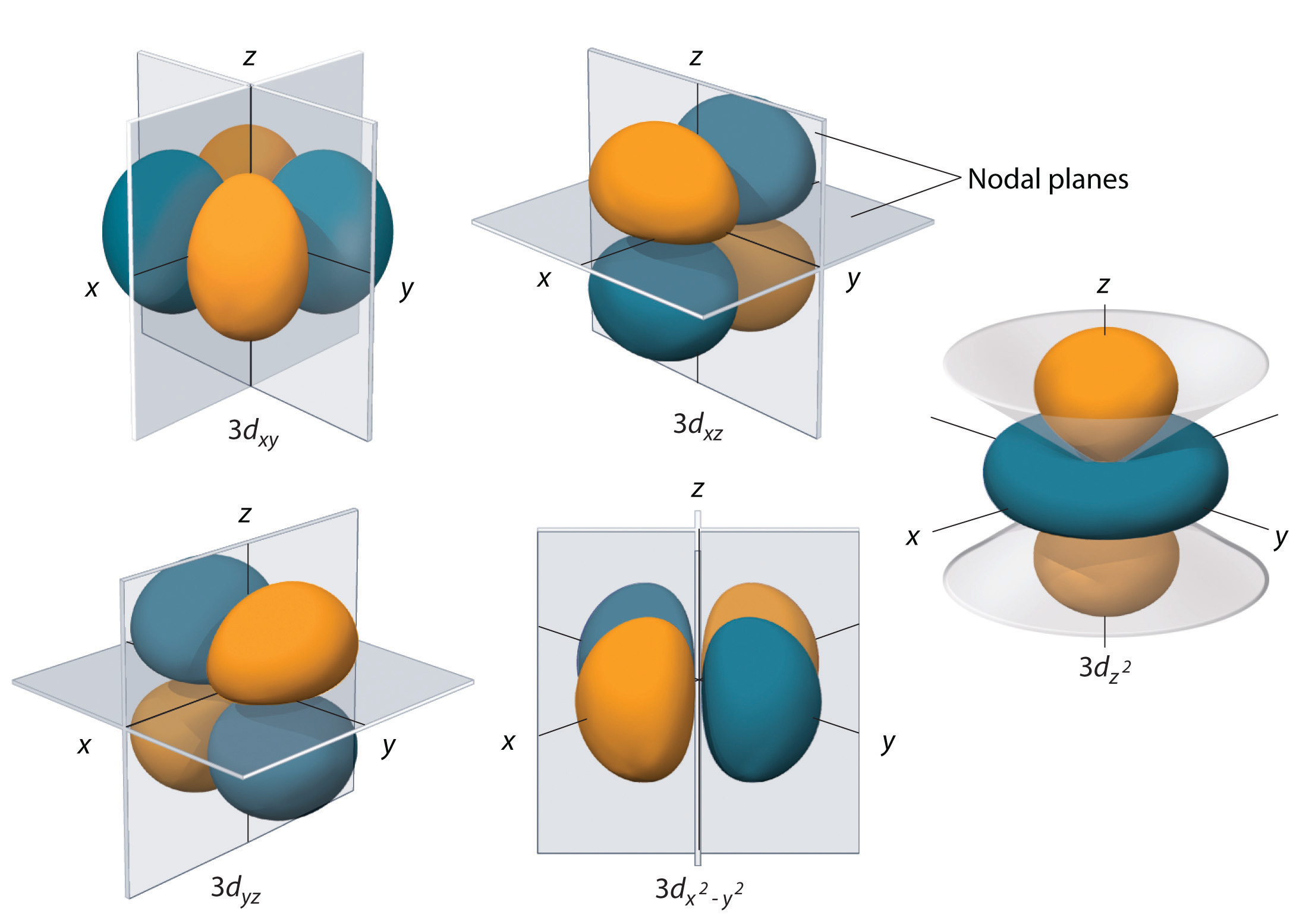
Hint: To answer this question you must be familiar with the concept of quantum numbers and the values that they take for a given orbital. An orbital diagram or electron shell diagram, which shows the arrangement of. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
